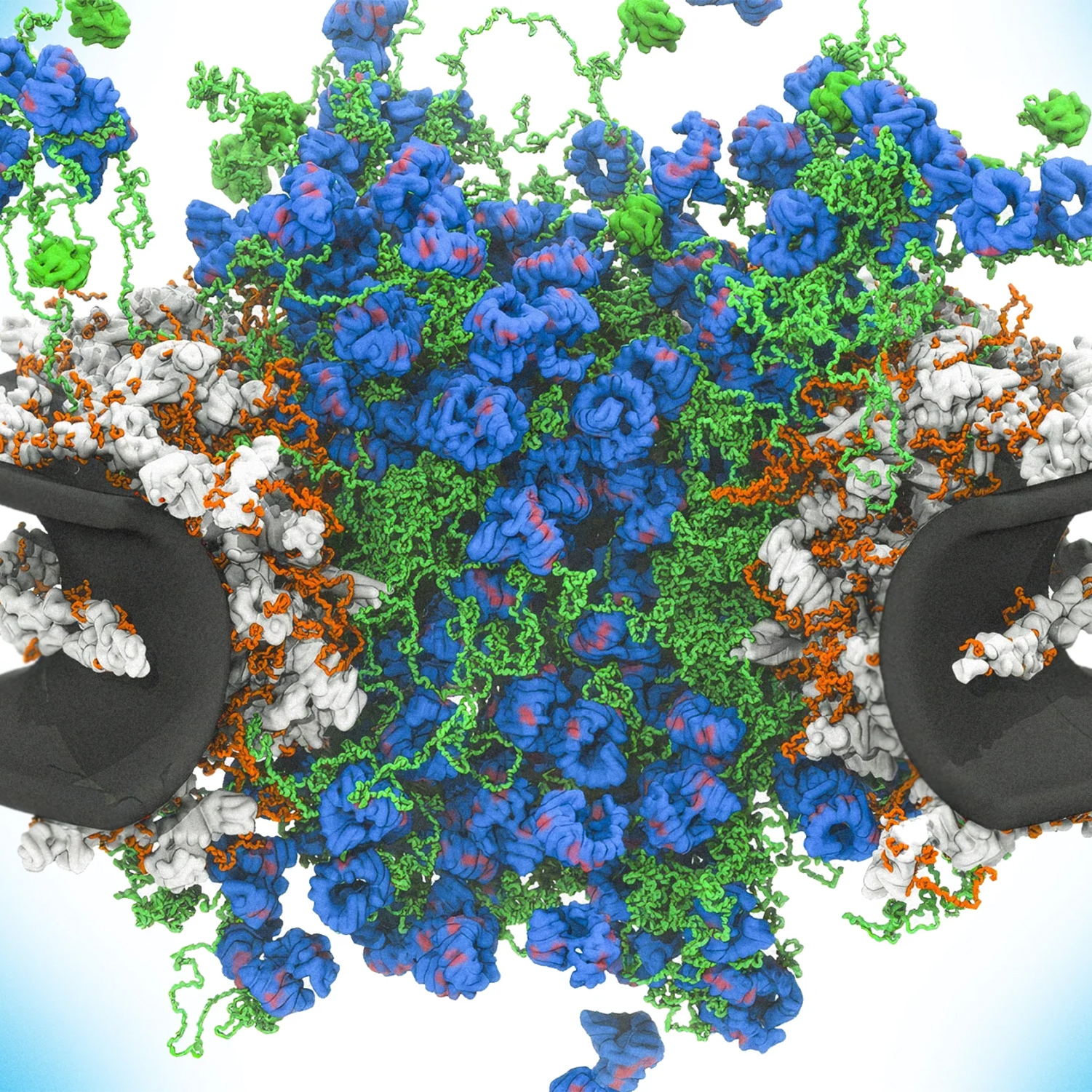
The nuclear pore complex serves as a critical, highly selective gatekeeper within eukaryotic cells, regulating the transport of molecules between the nucleus and the cytoplasm. Composed of hundreds of proteins, this complex utilizes intrinsically disordered proteins—often described as "wiggly" or "brush-like"—to create a dynamic, constantly rearranging channel. Recent high-speed atomic force microscopy reveals that transport proteins, known as karyopherins, actively interact with these disordered proteins to ferry cargo while simultaneously pushing away unauthorized molecules. This mechanism functions akin to a crowded dance floor, where only specific molecules possess the necessary "dance" to navigate the turbulent, fluctuating interior. Beyond its fundamental role in cellular health, the complex represents a potential point of vulnerability, as viruses and cancer cells may interfere with these dynamics to subvert normal cellular processes and evade immune responses.
Sign in to continue reading, translating and more.
Continue